Dose Reductions
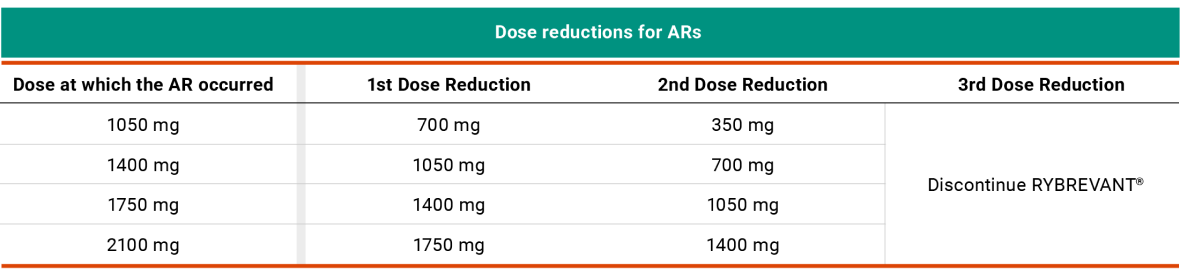
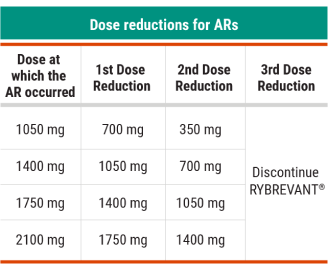
Recommended RYBREVANT® Dose Reductions for ARs1
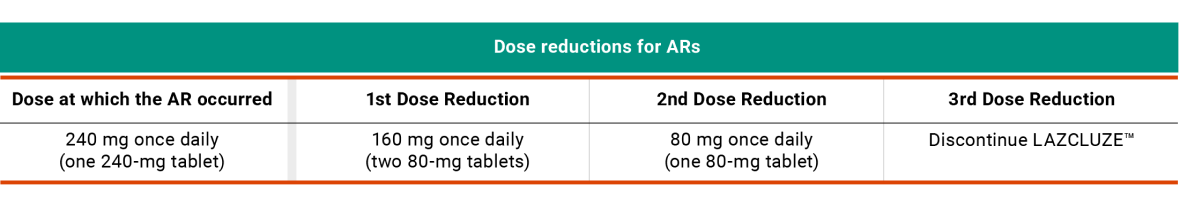
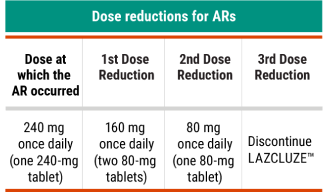
Recommended LAZCLUZE™ Dose Reductions for ARs2
Recommended LAZCLUZE™ Dose Reductions for ARs2
Adverse Event Severity Scale3
Based on the Common Terminology Criteria for Adverse Events (CTCAE) v5.0*
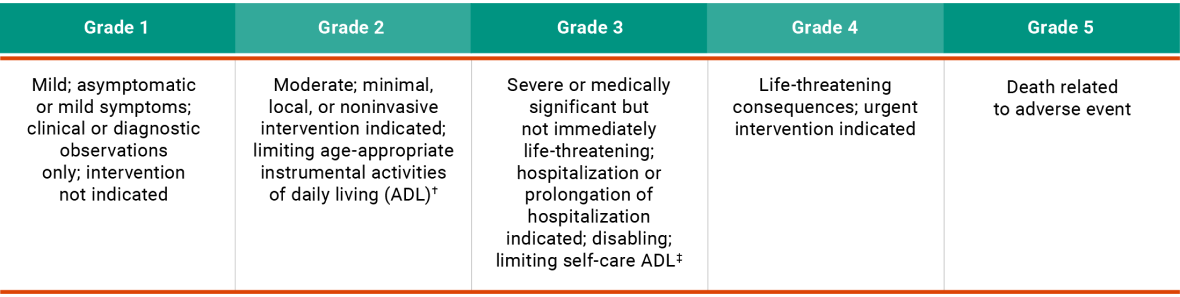

CTCAE definition may differ from the Prescribing Information.
Instrumental ADL refer to preparing meals, shopping for groceries or clothes, using the telephone, managing money, etc.
Self-care ADL refer to bathing, dressing and undressing, feeding self, using the toilet, taking medications, and not bedridden.
Dosage Modifications
Recommended dosage modifications for ARs1,2
Recommended dosage modifications for ARs for RYBREVANT® + LAZCLUZE™
For RYBREVANT® + LAZCLUZE™ refer to both the RYBREVANT® and LAZCLUZE™ recommendations. For RYBREVANT® + chemotherapy or RYBREVANT® as a single agent, refer only to the RYBREVANT® recommendations.


Recommended Dosage Modifications for ARs for RYBREVANT® in Combination With LAZCLUZE™1
When administering RYBREVANT® in combination with LAZCLUZE™, if there is an AR requiring dose reduction after withholding treatment and resolution, then reduce the dose of RYBREVANT® first.
Recommended Dosage Modifications for ARs for RYBREVANT® in Combination With Carboplatin and Pemetrexed1
When administering RYBREVANT® in combination with carboplatin and pemetrexed, modify the dosage of one or more drugs. Withhold or discontinue RYBREVANT® as shown in the table above. Refer to Prescribing Information for carboplatin and pemetrexed for additional dosage modification information.
AR, adverse reaction; ILD, interstitial lung disease; IRR, infusion-related reaction; TEN, toxic epidermal necrolysis; VTE, venous thromboembolism.
References:
- RYBREVANT® [Prescribing Information]. Horsham, PA: Janssen Biotech, Inc.
- LAZCLUZE™ [Prescribing Information]. Horsham, PA: Janssen Biotech, Inc.
- US Department of Health and Human Services. National Cancer Institute. Common terminology criteria for adverse events (CTCAE). Version 5.0. Published November 27, 2017. Accessed June 7, 2024. https://ctep.cancer.gov/protocoldevelopment/electronic_applications/docs/CTCAE_v5_Quick_Reference_5x7.pdf


